
Khoom Taw qhia
| Hypophosphorous acid Basic ntaub ntawv |
| Cov khoom siv tshuaj lom neeg siv tshuaj lom neeg muaj kev phom sij IDENTIFICATION Kev Npaj Txoj Cai Ntau Lawm |
| Khoom npe: | Hypophosphorous acid |
| Synonyms: | HYPOPHOSPHORUSACID, 30% (W / V) SOLUTION; HYPOPHOSPHORUSACID, 50% (W / V) SOLUTION; Hypophosphorus acid: (Phosphinic acid); Hypophosphorus acid, 50% w / w aq. Soln.; Hypophosphorous; Hypophosphorousacid (corrosiveliquid, acidic, inorganic, nos); Hypophosphorousacid, 50% w/waq.soln.; HYPOPHOSPHORUS ACID SOLUTION 50% AQUEOUS |
| CAS: | 6303-21-5 |
| MF: | HLO 2 P |
| MW: | 63.980501 |
| EINECS: | 228-601-5 |
| Khoom Categories: | HPA; Inorganics; 6303-21-5 |
| Mol File: | 6303-21-5.mol |
 |
|
| Hypophosphorous acid Chemical Properties |
| Melting point | -25 degree |
| Boiling point | 108 degree (759.8513 mmHg) |
| ceev | 1.206 g / mL ntawm 20 degree (lit.) |
| vapor siab | <17 mmHg ( 20 °C) |
| cia temp. | tsis muaj kev txwv. |
| solubility | Soluble hauv H2O, ethanol, ethyl ether |
| pka ua | pK{0}}.1. |
| daim ntawv | hygroscopic crystals los yog tsis muaj xim oily kua |
| xim | Tsis muaj xim |
| Dej Solubility | SOLUBLE |
| Merck | 13,4894 |
| Kev ruaj ntseg: | ruaj khov. Incompatible nrog cov hauv paus muaj zog. Reacts hnyav nrog oxidizing tus neeg sawv cev, muaj zog hauv paus, mercury (II) nitrate thiab mercury (II) oxide. Tsis txhob kub tshaj 100 C. |
| InChIKey | GQZXNSPRSGFJLY-UHFFFAOYSA-N |
| CAS DataBase Reference | 6303-21-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Hypophosphorous acid (6303-21-5) |
| EPA Tshuaj Registry System | Phosphinic acid (6303-21-5) |
| Cov ntaub ntawv kev nyab xeeb |
| Hazard Codes | C |
| Risk Statements | 34 |
| Cov lus hais txog kev nyab xeeb | 26-36/37/39-45 |
| RIDADR | UN 3264 8/PG 3 |
| WGK Germany | 1 |
| RTECS | SZ6400000 |
| TSCA | Yog lawm |
| HazardClass | 8 |
| PackingGroup | II |
| HS Code | 28111990 |
| Cov Ntaub Ntawv Muaj Xwm Ceev | 6303-21-5(Cov ntaub ntawv muaj phom sij) |
| MSDS Cov Ntaub Ntawv |
| Tus kws kho mob | Lus |
|---|---|
| Phosphinic acid | Lus Askiv |
| SigmaAldrich | Lus Askiv |
| ACROS | Lus Askiv |
| ALFA | Lus Askiv |
| Hypophosphorous acid Siv thiab Synthesis |
| Cov ntsiab lus | Hypophosphorous acid tseem hu ua "hypophosphite" Nws yog cov roj tsis muaj xim lossis deliquescence siv lead ua, nws yog ib qho tseem ceeb ntawm cov khoom zoo. Kev siv tseem ceeb yog txo tus neeg sawv cev rau electroless plating, phosphoric tiv thaiv tsis muaj xim ntawm cov resins, nws kuj tuaj yeem siv rau hauv cov tshuaj tiv thaiv esterification catalyst, cov tub yees, tshwj xeeb tshaj yog rau cov khoom siv purity siab sodium hypophosphite. Muaj ntau txoj hauv kev rau kev npaj, cov txheej txheem kev lag luam uas tsim tau yog ion exchange resin method thiab electrodialysis method. Cov khoom siv tshuaj lom neeg ntawm hypophosphorous acid, kev siv, tshuaj lom neeg, thiab cov txheej txheem tsim khoom raug kho los ntawm andy ntawm Chemicalbook. (2016-12-04) |
| Cov khoom siv tshuaj | Nws yog deliquescent crystals los yog tsis muaj xim roj. Melting point: 26.5 degree. Tus txheeb ze ntom ntom (qhov tshwj xeeb lub ntiajteb txawj nqus): 1.439 (khoom, 19 degree). Nws yog soluble hauv dej, ethanol thiab ether, thiab nws tuaj yeem sib xyaw hauv txhua qhov kev faib ua ke nrog dej, ethanol, acetone. Nyob rau hauv cov huab cua, nws yooj yim deliquesce rau syrupy kua, thiab cov aqueous kua yog acidic. Hypophosphorous acid yog monobasic acid, nyob rau hauv aqueous tov, Hypophosphorous acid yog muaj zog acid, Ka=10-2 (25 degree); nws yog qhov ruaj khov ntawm chav tsev kub; Disproportionation cov tshuaj tiv thaiv tuaj yeem tshwm sim ntawm 130 degree, decompose rau hauv phosphine thiab phosphorous acid: 2H3PO2=H3PO4+PH3 Nws muaj zog txo, hnyav hlau ntsev tov tuaj yeem rov qab rau cov hlau xws li Cu2 +, Hg2 +, Ag +, xws li: 4Ag+H3PO2+2H2)=4Ag+H3PO4+4H+ Nws tsis muaj zog oxidizer, nws tuaj yeem txo qis rau phosphine, phosphine thaum ntsib tus neeg sawv cev muaj zog. |
| Siv | 1. Hypophosphorous acid yog siv los txo tus neeg sawv cev rau electroless plating; 2. Nws tuaj yeem siv los tiv thaiv cov xim ntawm phosphoric acid resin; 3. Nws yog siv los ua esterification catalyst, cov tub yees; 4. Nws yog siv los tsim hypophosphite, sodium ntsev, manganese ntsev, hlau ntsev feem ntau yog siv los ua khoom noj khoom haus; 5. Hypophosphorous acid yog siv nyob rau hauv cov tshuaj thiab raws li txo tus neeg sawv cev, kev txiav txim ntawm arsenic, tellurium thiab sib cais ntawm tantalum, niobium thiab lwm yam reagents. 6. Nws muaj zog txo tus neeg sawv cev, Nws tuaj yeem siv rau kev npaj ntawm sodium hypophosphite, calcium phosphate thiab lwm yam hypophosphite. 7. Nws tuaj yeem siv rau lub plating da dej. Pharmaceuticals. txo tus neeg sawv cev. general reagents. 8. Nws muaj zog txo tus neeg sawv cev, nws tuaj yeem siv ua sodium hypophosphite, calcium phosphate thiab lwm yam hypophosphite. 9. Cov khoom no yog dav siv los txo tus neeg sawv cev, Ag, Cu, Ni, Hg thiab lwm yam hlau raug txo rau cov hlau sib xws, rau kev txheeb xyuas ntawm As, Nb, Ta thiab lwm yam reagents, nws tuaj yeem siv rau kev npaj ntawm Na. , K, Ca, Mn, Fe thiab lwm hom hypophosphite. |
| Toxicity | Nws yog non-combustible. Tab sis thaum sib cuag nrog lub qhov H tus neeg sawv cev, nws yuav ua rau hluav taws. Thaum ntsib tus neeg sawv cev oxidizing, kev tawm tsam hnyav thiab kev sib txuas tuaj yeem ua mus. Thaum nws yog cua sov kom siab, nws tuaj yeem decompose rau hauv cov pa roj phosphine heev, lossis txawm tawg. Nws yog corrosive. Hypophosphorous acid feem ntau ntxiv rau hauv cov dej qab zib, thiab vim nws tsis nqus. Yog li qhov kev pheej hmoo me me, tab sis tshwj xeeb tshaj yog muaj zog hypophosphite mob plab hnyuv. Tsis ntev nws txaws rau hauv lub qhov muag los yog sib cuag ntawm daim tawv nqaij, dej ntau yog siv los ntxuav. Cov neeg ua haujlwm tsim khoom yuav tsum hnav khaub ncaws tiv thaiv thiab lwm yam khaub ncaws tiv thaiv. Cov khoom siv ntau lawm yuav tsum raug kaw, rhiav yuav tsum tau ua kom cua zoo. |
| TSEEM CEEB | Nqe lus piav txog Hazard: Ua rau tawv nqaij kub hnyiab thiab qhov muag puas. Ua rau qhov muag puas loj Cov lus ceeb toom: Tsis txhob ua pa plua plav/fume/gas/mist/vapors/spray. Ntxuav kom huv si tom qab tuav. Hnav cov hnab looj tes tiv thaiv thiab tiv thaiv qhov muag / ntsej muag. Yog SWALLOWED: Yaug qhov ncauj. Tsis txhob ua rau ntuav. YOG NTAWM SKIN (los yog plaub hau): Tshem tawm / Tshem tawm tag nrho cov khaub ncaws uas tsis huv. Yaug daim tawv nqaij nrog dej / da dej. YOG HAUV EYES: Yaug ceev faj nrog dej rau ob peb feeb. Tshem cov looj ntsej muag, yog tias tam sim no thiab yooj yim ua. Txuas ntxiv yaug. YOG INHALED: Tshem tawm cov neeg raug tsim txom los ntawm huab cua ntshiab thiab so hauv qhov chaw xis kom ua pa. Tam sim ntawd hu rau POISON CENTER lossis tus kws kho mob/tus kws kho mob. Lub khw kaw lawm. Pov tseg cov khoom no thiab nws lub thawv rau cov khoom pov tseg los yog qhov chaw tshwj xeeb pov tseg. |
| Txoj kev npaj | 1. Phosphorus thiab barium hydroxide tov yog rhuab, barium ntsev Ba (H2PO2) 2 • 2H2O tuaj yeem tsim tau, sulfuric acid yog ntxiv rau hauv hypophosphorous acid barium tov, Ba2+ tuaj yeem ua dej nag: Ba(H2PO2)2+H2SO4=BaSO4+2H3PO2 Hypophosphorous acid tuaj yeem tau txais los ntawm evaporating nyob rau hauv qhov txo qis thiab qis kub crystallization. Vim nyob rau hauv cov txheej txheem no, lub solubility ntawm barium ntsev yog me me, yog li cov concentration ntawm tau txais Hypophosphorous acid tsis siab, cov khoom lag luam yuav tsum tau purified los ntawm recrystallization. 2. cov barium oxide (los yog txiv qaub) thiab cov tshuaj dawb phosphorus yog rhuab ua ke los tsim cov theem nrab barium phosphate (los yog calcium), thiab ces reacts nrog sulfuric acid, nws yog lim, concentrated kom tau cov khoom, los yog sodium hypophosphite tov pib H- hom ion txauv resin tuaj yeem muab cov khoom lag luam. Txoj kev no yuav tsum muaj ntau cov resin, thiab resin regeneration thiab ntxuav cov kauj ruam yog cumbersome, nws feem ntau raug nqi ntau tshaj $ 7 ib phaus, nws tsuas yog haum rau me me batch ntau lawm, thiab tsis haum rau loj-scale industrial daim ntaub ntawv. 3. Hypophosphorous acid yog npaj los ntawm electrodialysis txoj kev, nyob rau hauv lub electrodialysis cell faib mus rau hauv peb qhov chaw, lawv yog anode chamber, raw khoom chamber thiab cathode chamber, lub intermediate yog sib cais los ntawm anionic membrane thiab cationic membrane, ntawm ob daim nyias nyias sodium hypophosphite tov yog muab tso rau. (concentration ntawm 100g / L ~ 500g / L), anode chamber yog dilute tov ntawm Hypophosphorous acid 5g / L, anode chamber yog dilute sodium hydroxide tov (5g / L), nruab nrab ntawm tus ncej DC (3V ~ 36V) yog dhau, anode tso tawm oxygen, thiab tsim cov khoom thib ob ntawm Hypophosphorous acid; cathode emits hydrogen, thiab tsim cov khoom thib ob ntawm sodium hydroxide, lub sij hawm cov tshuaj tiv thaiv yog 3 ~ 21h. Cov tshuaj tiv thaiv ntawm anode chamber thiab cathode chamber yog raws li nram no: anode chav: H2O==H++OH- 2OH-==O2+2H2O+4e H++H2PO2-==H3PO2 Cathode Chamber: H2O==H++OH- 2H++2e==H2 Na++OH-==NaOH Electrodialysis txoj kev npaj Hypophosphorous acid yog qhov yooj yim thiab cov cuab yeej siv peev me me, nws tsim nyog rau ntau lawm. 4. Pib los ntawm qib qib sodium hypophosphite, Cl-, SO42-anions uas cuam tshuam rau cov ntsuas zoo ntawm Hypophosphorous acid raug tshem tawm los ntawm nag lossis daus, hnyav hlau ions raug tshem tawm ntawm kev daws los ntawm kev tsim sulfide, thiab tom qab ntawd siv cov kua qaub muaj zog. cation txauv resin kom tau txais sodium theem nrab phosphate, siab purity qib khoom tuaj yeem tau txais. Cov txheej txheem tuaj yeem tsim cov qib high-qib phosphate, technically yog ua tau, cov txheej txheem yog yooj yim, yooj yim lag luam, cov khoom zoo, nws muaj peev xwm ua tau raws li cov kev xav tau ntawm kev lag luam electronics, kev lag luam tiv thaiv thiab lwm yam high-tech teb. 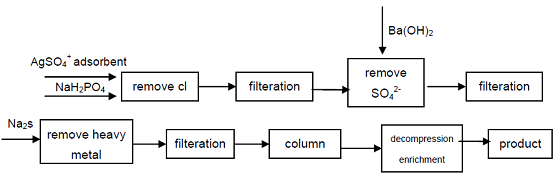 Daim duab 1 Cov Txheej Txheem Ntau Lawm ntawm Hypophosphorous Acid los ntawm Industrial Sodium Hypophosphite. 5. Ion txauv resin txoj kev: li 70g ntawm cation txauv resin wetted nrog dej yog ntim rau hauv lub khob raj nrog 5 mol / L hydrochloric acid circulating txog 15min, tom qab kom huv si ntxuav nrog dej, siab purity aqueous sodium hypophosphite aqueous tov (15 g / 60 ml H2O) ntws los ntawm nws, cov kab ke yog thawj zaug ntxuav nrog 50 ml, tom qab ntawd nrog 25 rnl distilled dej. Lub effluent acid thiab ntxuav yog ua ke, nws yog concentrated los ntawm evaporation nyob rau hauv dej da dej. Cov concentrated acid yog muab tso rau hauv lub tshuab nqus tsev siab nrog P205 tshuab ziab khaub ncaws rau lub cev qhuav dej, txias thiab crystallization, pom, recrystallization, kom tau txais cov khoom lag luam hypophosphorous acid. |
| Txoj kev tsim khoom | Ion txauv resin txoj kev: muab li 70 g dej-soluble cation txauv resins mus rau hauv lub khob raj. Ci nrog 5 mol / L hydrochloric acid rau li 15 feeb thiab ntxuav kom txaus nrog dej. Muaj cov tshuaj aqueous sodium hypophosphite (15 g / 60 ml H2O) kom ntws los ntawm kab ntawv cob, tom qab ntawd ntxuav thawj zaug nrog 50 ml dej, thiab tom qab ntawd yaug nrog 25 rnl dej distilled. Cov effluent acid thiab cov ntxhua khaub ncaws tau sib xyaw ua ke thiab ua kom muaj zog los ntawm evaporation ntawm lub da dej. Cov concentrated acid yog xa mus rau lub tshuab nqus tsev siab heev, P205 tshuab ziab khaub ncaws rau lub cev qhuav dej, ua raws li qhov txias crystallization, pom thiab recrystallization kom tau txais cov khoom tiav ntawm hypophosphorous acid. |
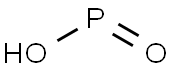
| Kev piav qhia | Hypophosphorous acid yog ib tug muaj zog txo tus neeg sawv cev nrog ib tug molecular mis ntawm H3PO2. Inorganic chemists xa mus rau cov kua qaub dawb los ntawm lub npe no txawm hais tias nws lub npe IUPAC yog dihydridohydroxidooxidophosphorus, los yog lub npe tau txais ntawm phosphinic acid. Nws yog ib qho xim tsis muaj xim uas tsis muaj melting, uas yog soluble hauv dej, dioxane, thiab cawv. Cov mis rau hypophosphorous acid feem ntau yog sau H3PO2, tab sis kev nthuav qhia ntau dua yog HOP (O) H2 uas qhia txog nws cov cim monoprotic. Cov ntsev tau los ntawm cov kua qaub no hu ua phosphinates (hypophosphites). |
| Kev piav qhia | Cov kua qaub no muaj cov qauv dav dav ntawm H4P2O6 thiab txawv ntawm lwm cov oxy-phosphorous acids. Nws muaj ntau yam peculiarities. Nws yog tsim nrog phosphorous thiab phosphoric acids, thaum phosphorus yog oxidized los ntawm huab cua noo. Yog tias dawb phosphorus raug huab cua, thiab sodium acetate tau ntxiv rau cov kua uas tsim, me ntsis insoluble sodium hypophosphate, Na2H2P2O6 · 6H2Oseparates. Txawm li cas los xij, sodium hypophosphate monohydrate yog soluble heev thiab deliquescent ntawm -98.7 g / 100 ml. |
| Chemical Properties | kua tsis muaj xim |
| Lub cev muaj zog | Tsis muaj xim deliquescent crystals los yog oily kua; qaub tsw; ceev 1.493 g / cm3; melts ntawm 26.5 degree; boils ntawm 130 degree; soluble heev nyob rau hauv dej, cawv thiab ether; den-sity ntawm 50% aqueous tov yog 1.13 g / mL. |
| Siv | Hypophosphorous acid feem ntau yog siv rau electroless nickel plating. Nws koom nrog hauv kev txo qis ntawm cov ntsev arenediazonium. Nws ua raws li ib qho ntxiv hauv Fischer esterification cov tshuaj tiv thaiv. Tsis tas li ntawd, nws ua haujlwm ua tus neeg sawv cev nruab nrab, antioxidant, catalyst hauv polymerization thiab poly condensation, thiab tus neeg sawv cev ntub dej. Tsis tas li ntawd, nws yog siv rau hauv kev tsim tshuaj, discoloration ntawm polymers, dej kho thiab retrieval ntawm precious los yog non-ferrous hlau. Ntxiv rau qhov no, nws yog siv los ua cov tshuaj bleaching rau plastics, hluavtaws fibers, decolorizing tus neeg sawv cev thiab rau xim stabilization thaum lub sij hawm tsim cov tshuaj thiab ntau yam yas. |
| Kev npaj | Hypophosphorous acid tuaj yeem npaj los ntawm ntau txoj hauv kev: 1. Boiling dawb phosphorus nrog calcium hydroxide: P4+ 4Ca(OH)2 + 8H2O → 4Ca(H2PO2)2 + 4H2 Calcium ntsev yog soluble hauv dej. Kev kho mob nrog sulfuric acid yields thehypophosphorous acid: (H2PO22 Ca + H2SO4 → 2H3PO2+ CASO4 Cov khoom sib tov yog lim kom tshem tawm cov CaSO4 insoluble. Lub aqueous solu-tion ntawm hypophosphorous acid yog concentrated nyob rau hauv txo lub siab.Concentrated baryta dej yuav siv tau es tsis txhob ntawm calcium hydroxide.2. Los ntawm kev kho sodium hypophosphite, NaH2PO2 nrog ib qho ion-exchange resin.Cov ntsev ntsev tuaj yeem tsim los ntawm kev npau npau dawb phosphorus nrog cov tshuaj sodium hydroxide, cov tshuaj tiv thaiv zoo ib yam li (1) saum toj no. PH3 + 2I2 + 2H2O → H3PO2+ 4HI Cov txheej txheem saum toj no tuaj yeem suav tias muaj kev nyab xeeb dua li qhov uas cuam tshuam nrog cua sov whitephosphorus nrog alkali. Hypophosphorous acid yuav tsum khaws cia hauv qab 50 degree. Nws yog muag commerciallyas ib tug aqueous tov ntawm ntau yam concentrations. |
| Txoj kev tsim khoom | Hypophosphorous acid yog tsim los ntawm cov tshuaj tiv thaiv ntawm barium hypophosphite thiab sulfuric acid, thiab lim tawm barium sulfate. Los ntawm evaporation ntawm cov tshuaj hauv lub tshuab nqus tsev ntawm 8 0 degree, thiab tom qab ntawd txias rau 0 degree, hypophosphorous acid crystallizes. |
| Txhais | ChEBI: Ib qho phosphorus oxoacid uas muaj ib qho pentavalent phosphorus covalently khi ntawm ib daim ntawv cog lus rau ob pawg hydrogens thiab ib pawg hydroxy thiab ntawm ob daim ntawv cog lus rau oxygen. Cov niam txiv ntawm chav kawm ntawm phosphinic acids. |
| Txhais | Ib tug dawb crystalline khoom. Nws yog monobasic acid tsim cov anion H2PO2- hauv dej. Cov ntsev ntsev, thiab yog li cov kua qaub, tuaj yeem npaj los ntawm cua sov daj phosphorus nrog sodium hydroxide tov. Cov kua qaub dawb thiab nws cov ntsev yog cov muaj zog txo cov neeg ua haujlwm. |
| Cov tshuaj tiv thaiv | Hypophosphorous acid yog miscible nrog dej nyob rau hauv tag nrho cov proportions thiab ib tug coj mus muag zog yog 30% H3PO2. Hypophosphites yog siv hauv tshuaj. Hypophosphorous acid yog ib tug haib txo tus neeg sawv cev, piv txwv li, nrog tooj liab sulfate cov ntaub ntawv cuprous hydride Cu2H2, xim av precipitate, uas evolves hydrogen gas thiab tawm tooj liab rau ua kom sov; nrog nyiaj nitrate yields finely faib nyiaj; nrog sulfurous acid yields sulfur thiab ib co hydrogen sulfide; nrog sulfuric acid yields sulfurous acid, uas reacts li saum toj no; Manganate tam sim ntawd nrog permanganate. |
| Kev piav qhia dav dav | Hypophosphorous acid zoo nkaus li tsis muaj xim oily kua los yog deliquescent crystals nrog ib tug qaub tsw. Qhov ceev 1.439 g / cm3. Melting point 26.5 degree. Inhalation ntawm vapors irritates los yog burns lub pa ib ntsuj av. Cov kua thiab vapors tuaj yeem ua rau khaus lossis hlawv qhov muag thiab tawv nqaij. |
| Cua & Dej Reactions | Deliquescent. Dej soluble. |
| Reactivity Profile | HYPOPHOSPHORUS ACID decomposes thaum rhuab rau hauv phosphoric acid thiab spontaneously flammable phosphine. Yog oxidized los ntawm sulfuric acid nrog kev tso tawm ntawm sulfur dioxide thiab sulfur. Reacts explosively nrog mercury(II) oxide [Meller, 1940, Vol. 4, 778 ib. Reacts hnyav nrog mercury(II) nitrate [Meller, 1940, Vol. 4, 993 ib. Neutralizes hauv paus hauv exothermic cov tshuaj tiv thaiv. |
| Hazard | Hluav taws kub thiab tawg muaj feem cuam tshuam nrog oxidizing cov neeg ua haujlwm. |
| Health Hazard | TOXIC; nqus tau, noj los yog tawv nqaij ntawm cov khoom yuav ua rau mob hnyav lossis tuag taus. Kev sib cuag nrog cov khoom molten yuav ua rau mob hnyav rau ntawm daim tawv nqaij thiab qhov muag. Tsis txhob sib cuag ntawm daim tawv nqaij. Cov teebmeem ntawm kev sib cuag lossis nqus pa tuaj yeem ncua sijhawm. Hluav taws yuav ua rau khaus, corrosive thiab/los yog cov pa phem. Cov dej ntws los ntawm kev tswj hluav taws los yog cov dej dilution tuaj yeem ua rau corrosive thiab / los yog tshuaj lom thiab ua rau muaj kuab paug. |
| Hluav Taws Xob | Tsis-combustible, tshuaj nws tus kheej tsis hlawv tab sis yuav decompose raws li cua sov los tsim corrosive thiab/los yog tshuaj lom pa taws. Ib txhia yog oxidizers thiab tej zaum yuav ignite combustibles (ntoo, ntawv, roj, khaub ncaws, thiab lwm yam). Kev sib cuag nrog cov hlau tuaj yeem hloov pauv cov pa hluav taws xob hydrogen. Cov thawv ntim khoom tuaj yeem tawg thaum cua sov. |
| Txoj Kev Purification | Phosphorous acid yog ib hom kab mob ntawm kev lag luam 50% hypophosphorous acid. Jenkins thiab Jones [J Am Chem Soc 74 1353 1952] purified cov ntaub ntawv no los ntawm evaporating txog 600mL nyob rau hauv 1L lub hwj ntawm 40o, nyob rau hauv txo siab (hauv N2), mus rau ib tug ntim ntawm txog 300mL. Tom qab cov tshuaj tau txias lawm, nws raug xa mus rau lub qhov ncauj dav Erlenmeyer lub hwj uas tau nres thiab sab laug hauv lub Dry-ice / acetone da dej rau ob peb teev kom khov (yog tias tsim nyog, nrog khawb ntawm phab ntsa). Thaum lub khob ntim tau tso rau ntawm ca 5o rau 12 teev, txog li 30-40% ntawm nws liquefied, thiab rov lim dua. Cov txheej txheem no tau rov ua dua, tom qab ntawd cov khoom tau muab khaws cia tshaj Mg (ClO4) 2 hauv lub tshuab nqus tsev desiccator hauv qhov txias. Tom qab crystallisations los ntawm n-butanol los ntawm dissolving nws nyob rau hauv chav tsev kub thiab ces txias nyob rau hauv ib tug dej khov-ntsev da dej ntawm -20o tsis tshwm sim mus purify nws ntxiv. Cov kua qaub dawb tsim deliquescent crystals m 26.5o thiab soluble hauv H2O thiab EtOH. NaH2PO2 ntsev tuaj yeem ntxuav tau los ntawm kev sib pauv anion resin [Klement Z Anorg Allgem Chem 260 267 1949.] |
| Hypophosphorous acid Kev npaj cov khoom thiab cov ntaub ntawv raw |
| Cov khoom nyoos | AMBERLITE(R) IRC -50 |
| Cov khoom npaj | XANTHURENIC ACID-->3,5-diisopropylphenol-->cmtirust agent T-708-->Sodium hypophosphite-->TRIS(2,2'-BIPYRIDYL)RUTHENIUM(II) CHLORIDE HEXAHYDRATE-->4-IODOPYRIDINE-2-CARBOXYLIC ACID-->2,3,5,6-Tetrafluorophenol-->Benzothiazole, 4-methyl- (7CI,8CI,9CI)-->BEHENIC ACID METHYL ESTER-->Phosphorous acid |
Cim npe nrov: Tuam Tshoj hypophosphorous acid manufacturers, lwm tus neeg, Hoobkas
Tug: Potassium Bromate
Tom ntej: Fluoroboric Acid
Koj Tseem Yuav Zoo Li
Xa kev nug



